 In Jennings RC, Zucchelli G, Ghetti F and Colombetti G (eds) Light as an Energy Source and Information Carrier in Plant Physiology. Proc Natl Acad Sci USA 96: 10056–10061īorisov AY (1996) Specific features of excitation migration in photosynthesis. īassi R, Croce R, Cugini D, and Sandonà D (1999) Mutation analysis of an higher plant antenna protein provides identification of chromophores bound into multiple sites. With respect to analogous determinations reported in the literature for organic solvents, this information is valuable for further understanding the in-protein chlorophyll excited states and excited state dynamics: in particular, for the calculation of Förster transfer rates by means of chlorophyll–chlorophyll overlap integral employing the Stepanov relation for emission and single chromophore transition energies according to the results of mutational analysis of chlorophyll binding sites. A functional relation of their absorption was given by Gaussian subband decomposition covering the entire Q x and Q y optical region together with the absolute value of the molar extinction coefficient. Difference absorption spectroscopy with respect to the corresponding proteins binding the complete pigment complement yielded the spectral shape and extinction of single chlorophyll a and b. Recombinant CP29 and LHC II antenna proteins missing individual chromophores were obtained by over-expression in bacteria without any changing of the primary protein sequence and in vitro reconstitution. 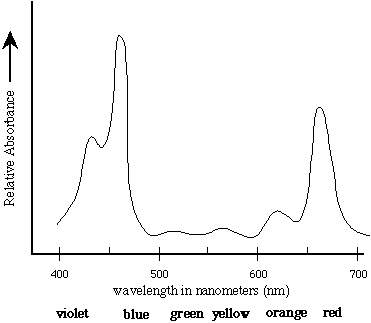
The spectral forms of the two chlorophyll species in higher plant Photosystem II antenna proteins have been experimentally determined within their protein environment. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
